Structure of Atom Revision Notes: Chapter wise Highlighted Points for Quick Revision(One-shot) for Competitive Exams 2025.
Structure of Atom – Quick Revision Notes 2025 is your one-stop solution for mastering one of the most crucial chapters in Chemistry! This page provides chapter-wise key points, summarized formulas, and quick concept highlights for faster and smarter revision. Designed for NEET, JEE, and CBSE Class 11–12 students, these notes help you understand every fundamental idea – from subatomic particles, atomic models, quantum numbers, to electronic configuration and orbitals.
Our expert team has simplified the entire Structure of Atom chapter into concise, exam-focused notes with diagrams, solved examples, and important MCQs. Whether you’re preparing for competitive exams or school tests, this guide ensures complete clarity and confidence in concepts. It’s perfect for last-minute revision, helping students recall essential theories like Bohr’s Model, Heisenberg’s Principle, and de Broglie’s Hypothesis in minutes.

Structure of Atom – Quick Revision Notes 2025
Explore this page to strengthen your basics, clear your doubts, and score higher in exams. Stay ahead with well-organized, easy-to-understand content that brings chemistry alive!
1. Subatomic Particles
- Electron (e⁻):
- Discovered by J.J. Thomson (1897) – Cathode ray tube experiment.
- Charge: –1.6 × 10⁻¹⁹ C
- Mass: 9.1 × 10⁻³¹ kg
- Charge/mass ratio (e/m): 1.7588 × 10¹¹ C/kg
- Proton (p⁺):
- Discovered by E. Goldstein (Canal rays)
- Charge: +1.6 × 10⁻¹⁹ C
- Mass: 1.673 × 10⁻²⁷ kg
- Neutron (n⁰):
- Discovered by James Chadwick (1932)
- Charge: 0
- Mass: 1.675 × 10⁻²⁷ kg
Note:
Mass of p⁺ ≈ n⁰ >> e⁻
Important Question:
Q. Who discovered neutron? → James Chadwick, 1932
2. Atomic Models
(A) Thomson’s Model (Plum Pudding Model)
- Atom = sphere of +ve charge with electrons embedded.
- Fails to explain: nuclear stability, scattering experiment results.
(B) Rutherford’s α-Scattering Experiment (1911)
- Gold foil experiment → bombarded by α-particles (He²⁺).
Observations:
- Most α-particles passed undeflected → atom mostly empty space.
- Few deflected → positive charge concentrated in small nucleus.
- Very few bounced back → dense massive center (nucleus).
Conclusions:
- Nucleus at center, +ve charged.
- Electrons revolve around nucleus.
- Size of nucleus ≪ size of atom (~10⁻¹⁵ m vs 10⁻¹⁰ m).
Limitations:
Could not explain atomic stability (electrons should fall into nucleus).
3. Bohr’s Model (1913)
Postulates:
- Electron revolves in certain discrete orbits (stationary states) without radiating energy.
- Angular momentum quantization: mvr=(nh/2π), where n = 1, 2, 3, … (principal quantum number).
- Energy emission/absorption: occurs when electron jumps between orbits: ΔE=E2−E1=hν
- Energy of nth orbit: En=−13.6/n2 eV (for H atom)
- Radius of nth orbit: rn=0.529×10−10 × n2 m
Bohr’s Success:
✅ Explained Hydrogen spectrum
✅ Quantized energy levels
Limitations:
❌ Fails for multi-electron atoms
❌ Couldn’t explain Zeeman or Stark effects
❌ Violates uncertainty principle
PYQ (NEET/JEE):
Q. Calculate energy difference between n = 2 and n = 1 in hydrogen → ΔE = 10.2 eV
4. Electromagnetic Radiation & Spectrum
Electromagnetic Waves:
- Travel at speed of light, c = 3 × 10⁸ m/s
- Relation: c=λν where λ = wave length, ν = frequency
Energy of photon: E=hν=hc/λ (h = 6.626 × 10⁻³⁴ J·s)
Spectral Series of Hydrogen (Balmer Formula):
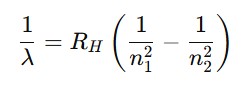
| Series | n₁ | n₂ Range | Region |
|---|---|---|---|
| Lyman | 1 | 2 → ∞ | UV |
| Balmer | 2 | 3 → ∞ | Visible |
| Paschen | 3 | 4 → ∞ | IR |
| Brackett | 4 | 5 → ∞ | IR |
| Pfund | 5 | 6 → ∞ | IR |
PYQ:
Q. Which series lies in visible region? → Balmer Series
5. Quantum Mechanical Model (QMM)
Developed by Schrödinger (1926) using wave mechanics.
Schrödinger Equation: Hψ=Eψ
ψ = wave function, ψ² = probability density of finding electron.
Heisenberg Uncertainty Principle: Δx⋅Δp≥(h/4π)
Cannot know both position & momentum of electron simultaneously.
Key Idea: Electron = wave + particle dual nature.
Wave–Particle Duality (de Broglie): λ=h/mv
PYQ:
Q. What is the de Broglie wavelength of an electron moving with 1×10⁶ m/s? → Apply λ = h/mv.
6. Quantum Numbers
- Principal Quantum Number (n):
- Orbit / energy level
- Values: 1, 2, 3, …
- Determines size, energy
- No. of orbitals in shell = n²
- Max electrons = 2n²
- Azimuthal (Angular) Quantum Number (l):
- Subshell type
- Values: 0 to (n–1)
- l = 0 → s
- l = 1 → p
- l = 2 → d
- l = 3 → f
- Shape: s = spherical, p = dumbbell, d = cloverleaf and f = complex multilobed shapes
- Magnetic Quantum Number (mₗ):
- Orientation of orbital
- Values: –l → +l (total 2l+1 orbitals)
- Spin Quantum Number (mₛ):
- +½ (clockwise), –½ (anticlockwise)
Examples:
- n = 2 → l = 0,1 → 2s, 2p → mₗ = 0 for s, –1,0,+1 for p → total 4 orbitals
Important Table:
| Shell | n | l | Subshell | Orbitals | Max e⁻ |
|---|---|---|---|---|---|
| K | 1 | 0 | 1s | 1 | 2 |
| L | 2 | 0,1 | 2s, 2p | 4 | 8 |
| M | 3 | 0,1,2 | 3s, 3p, 3d | 9 | 18 |
| N | 4 | 0,1,2,3 | 4s, 4p, 4d, 4f | 16 | 32 |
7. Rules for Filling of Electrons
(A) Aufbau Principle
Electrons fill orbitals in increasing energy order: 1s<2s<2p<3s<3p<4s<3d<4p<5s<4d<5p<6s<4f<5d<6p<7s…1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s …1s<2s<2p<3s<3p<4s<3d<4p<5s<4d<5p<6s<4f<5d<6p<7s…
(Energy order given by n + l rule)
If (n+l) same → lower n filled first.
(B) Pauli’s Exclusion Principle
No two electrons in an atom can have all four quantum numbers identical → ⇒ max 2 e⁻ per orbital, opposite spins.
(C) Hund’s Rule of Maximum Multiplicity
Electrons occupy degenerate orbitals singly before pairing up, with parallel spins.
Examples:
- Oxygen: 1s² 2s² 2p⁴ → ↑↓ ↑↓ ↑↓ ↑ ↑
- Nitrogen: 1s² 2s² 2p³ → ↑↑↑ (half-filled = stable)
Exceptional Configurations:
- Cr: [Ar] 3d⁵ 4s¹
- Cu: [Ar] 3d¹⁰ 4s¹
Reason: half/full-filled d orbital stability.
PYQ (NEET):
Q. Which has half-filled subshell? → Cr (Z=24)
8. Orbitals and Their Shapes
- s-orbital: spherical; one orientation
- p-orbitals: dumbbell-shaped; 3 orientations (pₓ, pᵧ, pz)
- d-orbitals: 5 orientations; clover-leaf shaped
- f-orbitals: complex; 7 orientations
Node Concept:
Total nodes = (n – 1)
Radial nodes = (n – l – 1)
Angular nodes = l
Example:
3p orbital → n = 3, l = 1
→ Radial nodes = 3–1–1=1
→ Angular nodes = 1
→ Total = 2
9. Photoelectric Effect (Einstein’s Equation)
Equation: hν=hν0 + ½ mv2
where hν₀ = work function, (1/2)mv² = kinetic energy of emitted electron.
Observations:
- No photoelectron below threshold frequency.
- K.E. depends on frequency, not intensity.
- No time lag between illumination & emission.
PYQ:
Q. Which property of light explains photoelectric effect? → Particle nature (photon concept)
10. Important Formula Collection
| Concept | Formula | Notes |
|---|---|---|
| Energy of nth orbit | Eₙ = –13.6/n² eV | Hydrogen only |
| Radius of nth orbit | rₙ = 0.529 × 10⁻¹⁰ × n² m | Bohr radius for H |
| Velocity of electron | vₙ = 2.18 × 10⁶ / n m/s | Decreases with n |
| Energy difference | ΔE = 13.6 (1/n₁² – 1/n₂²) eV | |
| Photon energy | E = hν = hc/λ | |
| de Broglie wavelength | λ = h/mv | |
| Momentum of photon | p = h/λ | |
| No. of orbitals per shell | n² | |
| Max electrons per shell | 2n² | |
| Radial nodes | n – l – 1 | |
| Angular nodes | l |
11. Previous Year Conceptual Hotspots
Hydrogen Spectrum (Bohr model)
- Lyman → UV
- Balmer → Visible
- Paschen, Brackett, Pfund → IR
→ Repeated in NEET almost every 2 years.
Quantum Numbers:
- Given (n, l ,m) values → identify orbital.
Ex: n=3, l=2 → 3d; m = –2 → one of 3d orbitals.
Magnetic Moment (for d-block): μ= √[n(n+2)] BM
where n = unpaired electrons.
Electron Configuration Stability:
- Cu, Cr, Mo, Ag, Au → exceptional.
Uncertainty & de Broglie Relation Questions
– conceptual + numerical.
Pauli, Hund, Aufbau combinations
– “Which configuration violates?” type.
Work Function & Threshold Frequency numericals.
Calculation of wavelength/energy difference between orbits.
Questions on total orbitals/electrons for given quantum numbers.
Identify wrong statement (Bohr or QMM postulates).
12. Concept Shortcards / Flash Reminders
Bohr’s Constant Energy Levels:
E₁ = –13.6 eV
E₂ = –3.4 eV
E₃ = –1.51 eV
E₄ = –0.85 eV
Photon Emission:
Energy emitted when e⁻ falls from n₂ → n₁ → difference in energy levels.
Example:
For He⁺ (Z=2):
r₁ = 0.265 Å; E₁ = –54.4 eV
Energy ∝ Z²/n²
13. Commonly Confused Facts
| Concept | Clarification |
|---|---|
| 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² | 4s filled before 3d |
| 3d orbitals start filling after 4s | Energy order based on n+l rule |
| For ions: remove electrons from outermost shell first | Ex: Fe²⁺ = [Ar] 3d⁶ (4s electrons lost first) |
| Orbit → path (Bohr); Orbital → region (QMM) | Distinguish |
| ψ² → probability density | Not charge density |
| Uncertainty applies for micro, not macro particles | Only for atomic scale |
14. Sample Tricky NEET/JEE Questions
Q1. Identify orbital with highest energy:
(A) 3p (B) 4s (C) 3d (D) 4p
→ Use (n+l) rule → 3p (3+1=4), 4s (4+0=4), smaller n wins → 3p < 4s
→ Highest = 4p
Q2. Number of radial nodes in 4d orbital → n=4, l=2 → 4–2–1=1
Q3. Calculate wavelength of light emitted for n₂=3 → n₁=2 transition in H atom.
→ Apply ΔE = 13.6(1/2² – 1/3²) eV → 1.89 eV → λ = 1240/1.89 ≈ 656 nm (Balmer series)
Q4. For Cr³⁺, e⁻ configuration → [Ar] 3d³
Q5. Which violates Pauli’s exclusion?
– Both electrons in 1s with same spin.
15. Miscellaneous Key Points
- Isotopes: Same Z, different A (¹H, ²H, ³H)
- Isobars: Same A, different Z (¹⁴C, ¹⁴N)
- Isotones: Same no. of neutrons (¹⁴C, ¹⁵N)
- Isoelectronic: Same e⁻ count (Na⁺, Ne, F⁻)
Electron energy in orbit: Negative (bound system).
Ground state: lowest energy level (n=1).
Ionization energy: energy to remove e⁻ from ground state (13.6 eV for H).
Nodal plane: plane where ψ = 0.
Visual Mnemonics
Energy order shortcut:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s
Spectral Series mnemonic:
“Lazy Boy Paints Beautiful Pictures” →
Lyman, Balmer, Paschen, Brackett, Pfund
17. Final 1-Day Rapid Review Table
| Topic | Key Point | Formula / Tip |
|---|---|---|
| Electron | Thomson (–), e/m = 1.7588×10¹¹ | |
| Proton | Goldstein (Canal rays) | |
| Neutron | Chadwick (1932) | |
| Bohr model | mvr = nh/2π | Eₙ = –13.6/n² |
| Spectrum | 1/λ = R(1/n₁²–1/n₂²) | Balmer → visible |
| de Broglie | λ = h/mv | Wave–particle duality |
| Uncertainty | Δx·Δp ≥ h/4π | Limits measurement |
| Quantum nos. | n, l, m, s | Defines orbital |
| Aufbau | (n+l) rule | Energy order |
| Pauli | Max 2e⁻ per orbital | Opp. spin |
| Hund | Degenerate → singly filled first | Parallel spin |
| Radial nodes | n–l–1 | |
| Angular nodes | l | |
| Energy gap (H) | 13.6(1/n₁²–1/n₂²) | eV |
Thank you for visiting our website! We’re committed to keeping your learning journey smooth and updated with the latest 2025 revision materials. Don’t forget to bookmark this page and return regularly for new notes, solved questions, and smart study tips. Please share this page with your friends and classmates so they can benefit too. If you have any questions or need more help, feel free to contact our support team anytime. Keep visiting, keep learning, and stay confident – your success in chemistry starts right here!